 Physicians in the United Kingdom will be able to legally prescribe medical marijuana products to their patients starting next month. The policy shift follows a public outcry over cases of young epileptic patients who were denied the only treatments that worked for them.
Physicians in the United Kingdom will be able to legally prescribe medical marijuana products to their patients starting next month. The policy shift follows a public outcry over cases of young epileptic patients who were denied the only treatments that worked for them.

 After years of upholding employee firings for use of cannabis even under state medical marijuana programs, the courts are finally starting to turn around on the question.
After years of upholding employee firings for use of cannabis even under state medical marijuana programs, the courts are finally starting to turn around on the question. Last month, the DEA enthused the pharmaceutical industry but disappointed cannabis advocates by re-scheduling the drug Epidiolex—but not CBD, the cannabinoid that makes it work. Now word emerges of a letter to the DEA by the Food & Drug Administration essentially calling for the descheduling of CBD altogether.
Last month, the DEA enthused the pharmaceutical industry but disappointed cannabis advocates by re-scheduling the drug Epidiolex—but not CBD, the cannabinoid that makes it work. Now word emerges of a letter to the DEA by the Food & Drug Administration essentially calling for the descheduling of CBD altogether.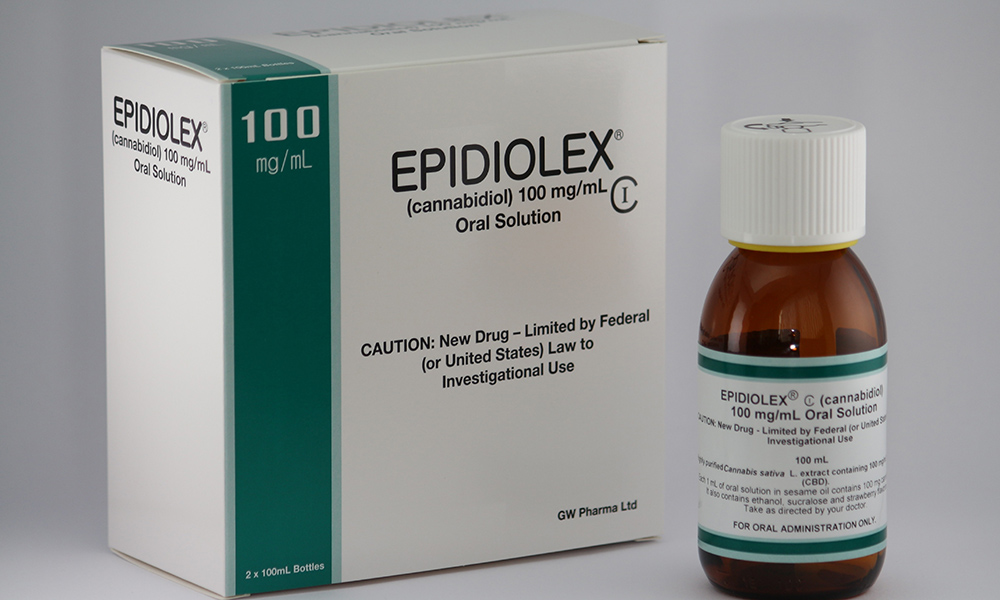 With the DEA's rescheduling of Epidiolex, shares in the British manufacturer of the drug are soaring. But CBD—the actual cannabinoid that the product is based on—is to remain in the restrictive Scheudle 1.
With the DEA's rescheduling of Epidiolex, shares in the British manufacturer of the drug are soaring. But CBD—the actual cannabinoid that the product is based on—is to remain in the restrictive Scheudle 1.
 Among several cannabis-related bills that cleared California's state house before the last legislative session came to close is one that would lift the tax burden on medical marijuana providers. The bill is intended to again open space for "compassionate care," which was ironically squeezed out under California's adult-use regulation regime.
Among several cannabis-related bills that cleared California's state house before the last legislative session came to close is one that would lift the tax burden on medical marijuana providers. The bill is intended to again open space for "compassionate care," which was ironically squeezed out under California's adult-use regulation regime.  Having just undertaken a "critical study" of CBD, the non-psychoactive cannabinoid held to have multifarious medicinal applications, the World Health Organization is now opening such a study on THC. Stigmatized due to its psychoactive properties and currently in the shadow of the suddenly sexy CBD, tetrahydrocannabinol shows its own potential for application by the medical industry.
Having just undertaken a "critical study" of CBD, the non-psychoactive cannabinoid held to have multifarious medicinal applications, the World Health Organization is now opening such a study on THC. Stigmatized due to its psychoactive properties and currently in the shadow of the suddenly sexy CBD, tetrahydrocannabinol shows its own potential for application by the medical industry.





Recent comments
5 weeks 4 hours ago
6 weeks 1 day ago
6 weeks 6 days ago
12 weeks 1 day ago
18 weeks 4 days ago
18 weeks 4 days ago
21 weeks 5 days ago
22 weeks 4 days ago
26 weeks 4 days ago
30 weeks 3 days ago